- Dharmaconスクリーニングライブラリー
- SMARTvector Inducible Lentiviral shRNAプール化ライブラリー
SMARTvector Inducible Lentiviral shRNAプール化ライブラリー
最適化された機能分析スクリーニングのための誘導発現型shRNA

信頼性の高い機能分析スクリーニングを得るための最適なshRNA発現
Horizonの最先端のshRNAプラットフォームであるSMARTvector inducible lentiviral shRNAsは、プロモーターおよびレポーター選択が柔軟であり、最先端の合理的な設計アルゴリズムと組み合わせたTet-On® 3Gトランスアクチベータータンパク質による厳格な発現調節を利用し、ヒト、マウス、およびラットのタンパク質コードおよび長い非コード遺伝子の完全なゲノムを標的としています。
プール化Lentiviralライブラリーは、多数の遺伝子をスクリーニングするための効率的で費用効果の高い方法を提供します。ライブラリーの構築、プーリング技術、およびハイスループットシーケンス対応のスクリーニングワークフローは、再現性と正確なヒット識別を確実にするために実験的に検証されています。プール化lentiviralスクリーニング用ライブラリーのページで、最適なライブラリー設計とプール化スクリーニングワークフローについて学ぶことができます。
Highlights:
- SMARTvector inducible lentiviral shRNAによる強力な誘導性shRNA発現のための革新的なベクター設計
- 実験で最適なパフォーマンスを得るために、4つのプロモーターと2つの蛍光レポーターを選択可能
- Tet-On® 3Gトランスアクチベータータンパク質による厳密に制御されたshRNA発現誘導
恒常的shRNA発現には、SMARTvector lentiviral shRNAプール化スクリーニング用ライブラリーを選択してください。
各SMARTvector inducible Lentiviral shRNAライブラリーには、以下が含まれます。
- レンチウイルス粒子
- 5000 コンストラクト以下の場合:ライブラリー総量200 µL (8 x 25 µL), 5 x 107 TU/mL ± 20%
- 5001 コンストラクト以上の場合:ライブラリー総量400 µL (16 x 25 µL), 5 x 107 TU/mL ± 20%
- 100のnon-targetingコントロールコンストラクト
- 遺伝子特異的コントロールとして34のヒトまたはマウスのタンパク質コード遺伝子を標的とする272(遺伝子あたり8つのshRNA)のコンストラクト(一般的なレファレンス遺伝子(PPIB, GAPD, Actin, Lamin)および生存関連遺伝子が含まれます)
- コンストラクト配列、ターゲット遺伝子ID、数百万のマッピングされたリードごとのカウントを含むデータファイル
ヒトおよびマウスについては、遺伝子ファミリーから全ゲノムまでの組み合わせ済みSMARTvector Inducible Lentiviral shRNAライブラリーご用意しています。ラット用のライブラリーおよびカスタムSMARTvector Inducible Lentiviral shRNAコレクションは、ご要望に応じてご用意可能です。
- shRNAのカスタムSMARTvector Inducible Lentiviral shRNAコレクション
- 50~10,000のコンストラクトのプールとしてご利用いただけます。
- 精製、濃縮されたレンチウイルス粒子としてご提供
- 誘導的発現ベクター力価:1 x 107 TU/mL以上
ご注文前に、以下の資料より、プール化Lentiviral shRNAスクリーニングを慎重に計画し、必要なコンポーネントの量を計算してください。
- SMARTvector & shMIMIC lentiviral pooled libraries technical manual
- Pooled lentiviral screening laboratory protocols & Calculation tracking worksheet
検証済みのプロトコールで必要な試薬:
- トランスダクションの最適化のための、適切なSMARTvector Inducible Non-targetingコントロール
- SMARTvector Indexing PCR and Sequencing Primer Kits (A & B) :12種類のユニークなインデックスプライマーを含み、以下のような実験に最適化され、実験的に検証されています。
- 最小限のバイアスでゲノムDNAを効率的にPCR増幅
- ヒット識別のための高スループット多重化次世代シーケンシング
MARTvector Inducible Lentiviral shRNAプールライブラリーカバレッジ *:
| Collection | Human | Mouse | ||
|---|---|---|---|---|
| # targeted genes | Number pools and average shRNA per pool | # targeted genes | Number pools and average shRNA per pool | |
| Genome | 19241 | 16 pools of 9513 shRNA | 21745 | 18 pools of 9502 shRNA |
| Druggable genome | 7341 | 6 pools of 9735 shRNA | 9723 | 8 pools of 9670 shRNA |
| GPCR | 377 | 1 pool of 3012 shRNA | 494 | 1 pool of 3909 shRNA |
| Ion Channel | 340 | 1 pool of 2709 shRNA | 332 | 1 pool of 2646 shRNA |
| Phosphatase | 245 | 1 pool of 1948 shRNA | 268 | 1 pool of 2141 shRNA |
| Protease | 466 | 1 pool of 3702 shRNA | 529 | 1 pool of 4206 shRNA |
| Protein kinase | 702 | 1 pool of 5602 shRNA | 697 | 1 pool of 5558 shRNA |
| Ubiquitin conjugation | 557 | 1 pool of 4431 shRNA | 511 | 1 pool of 4069 shRNA |
| Apoptosis | ** | |||
| Cell cycle regulation | ** | |||
| De-ubiquitinating enzymes | ** | |||
| Membrane trafficking | ** | |||
| DNA damage response | ** | |||
| Epigenetics | ** | |||
| Nuclear receptor | ** | |||
| Transcription factors | ** | |||
重要なお知らせ
本製品は、以下に記載されている封じ込め措置および適用される法律と規制が満たされている実験室での(製品の利用規約に記載されている)社内での使用のみを目的としています。製品は、診断、治療、またはその他の商業目的で使用することはできません。また、いかなる目的でもヒトに、治療目的で動物に投与することはできません。本製品は複製能力がなく、自己不活性化(SIN)であり、非病原性です(感染性のヒト疾患を引き起こしません)。
レンチウイルス粒子製品を購入する研究者は、レンチウイルスベクター粒子の取り扱いに関する特定のガイドラインについて、施設の健康およびバイオセーフティ担当者に相談する責任があります。さらに、各研究者は、研究に必要な許可を取得し、複製不適格なSINレンチウイルスベクターと複製欠陥のあるレンチウイルス粒子を現地の管轄区域と機関に受け入れる責任を負います。
SMARTvector Inducible shRNA Lentiviralベクターデザイン
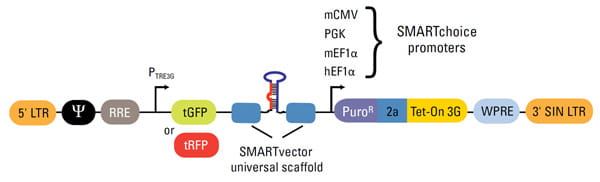
| Vector Element | Utility |
|---|---|
| 5' LTR | 5' Long Terminal Repeat necessary for lentiviral particle production and integration of the construct into the host cell genome |
| Ψ | Psi packaging sequence allows viral genome packaging using lentiviral packaging systems |
| PTRE3G | Inducible promoter with Tetracycline Response Element is activated by the Tet-On® 3G protein in the presence of doxycycline |
| tGFP or tRFP | TurboGFP or TurboRFP reporter for visual tracking expression upon doxycycline induction |
| SMARTvector universal scaffold | Optimized proprietary scaffold based on native primary microRNA in which gene-targeting sequence is embedded |
| PuroR | Puromycin resistance permits antibiotic selection of transduced cells |
| 2a | Self-cleaving peptide enables the expression of both PuroR and Tet-On® 3G transactivator from a single RNA pol II promoter |
| Tet-On® 3G | Encodes the doxycycline-regulated transactivator protein, which binds to PTRE3G promoter only in the presence of doxycycline |
| WPRE | Woodchuck Hepatitis Post-transcriptional Regulatory Element enhances transgene expression in target cells |
| 3' SIN LTR | 3' Self-inactivating Long Terminal Repeat for generation of replication-incompetent lentiviral particles |
単一ベクターにおける最適化された誘導的遺伝子ノックダウンシステム
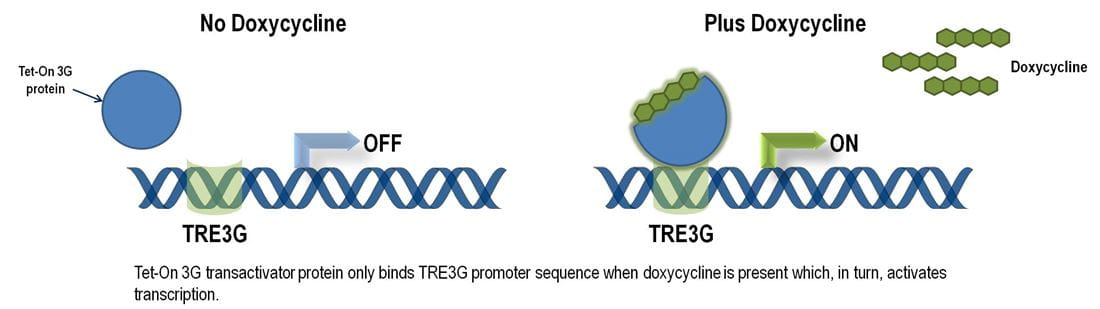
SMARTvector Inducible Lentiviral shRNAベクターは、Tet-On® 3G二部誘導システムを組み込んでいます。第3世代のTet誘導システムは、最小限の基礎発現(最小の漏れ)と誘導時の強力な活性化のために大幅に改善および最適化されています((Zhou X, et al., Gene Therapy 13, 1382 (2006)およびLoew R, et al., BMC Biotechnol. 10, 81 (2010))。Tet-On® 3G誘導システムにより、厳密に制御されたshRNA発現と、in vivoおよびin vitroでの遺伝子機能の研究が、前例のない精度で可能になります。
SMARTvectorプール化ライブラリーを用いたスクリーニングワークフロー
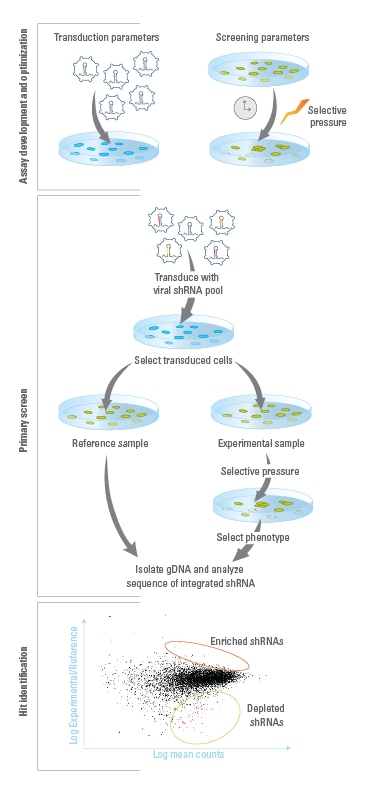
アッセイの開発と最適化:最適な実験条件を確立します。これには、a)レンチウイルスの形質導入、b)選択圧やアッセイ時間などのスクリーニングパラメーターが含まれます。一次スクリーニング:細胞当たり1つのshRNAコンストラクトを発現させるために、低MOIでレンチウイルスプールを細胞に形質導入します。レンチウイルスの感染した細胞を、対照用サンプルと選択圧をかける処理用サンプルに分けます。処理用サンプルに選択圧をかけた後、対照用サンプルおよび処理用サンプルの細胞それぞれからゲノムDNAを抽出します。イルミナに適合したプライマーとPhusion Hot-Start II High Fidelity DNA Polymeraseを使用して、ゲノム中に挿入されたshRNAコンストラクト配列をPCR増幅し、アダプター付加したPCR増幅断片をillumina flow cellに固定化します。結果的に生じるアンプリコンをIlluminaシークエンサーによりシークエンシングします。ヒットの識別とフォローアップ:コンストラクト配列を、対照用サンプルおよび処理用サンプルで識別します。スクリーン中に濃縮または枯渇したコンストラクトはヒットとして識別され、それらがターゲットとする遺伝子が識別されます。ヒットは、Dharmaconカタログコレクションから注文できる個別の構成要素を使用してさらに確認および調査できます。
SMARTvector Inducible Lentiviral shRNAプロモーター活性は細胞タイプによって異なる
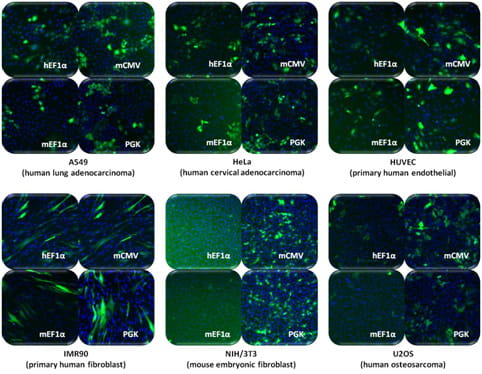
図中の細胞株に、SMARTvector Inducible Lentiviral shRNA Non-targetingコントロールをMOI = 0.3で形質導入しました。形質導入の24時間後、non-targeting shRNAおよびTurboGFPの発現を1 µg/mLドキシサイクリンで誘導しました。ドキシサイクリンの存在下で48時間培養した後、細胞をHoescht-33342で染色し、核(青)とTurboGFP(緑)を画像化しました。フィールド内の細胞の約30%が形質導入されていますが、特定の細胞タイプでは、恒常的プロモーター活性が低いことを示すTurboGFP発現が低いため、一部の画像には30%未満のTurboGFP陽性細胞が含まれているように見える場合があります。
SMARTvector Inducible Lentiviral shRNAは、4つのプロモーターオプションすべての使用において、ヒト初代細胞の特定の標的遺伝子の調整可能なノックダウンを可能にする
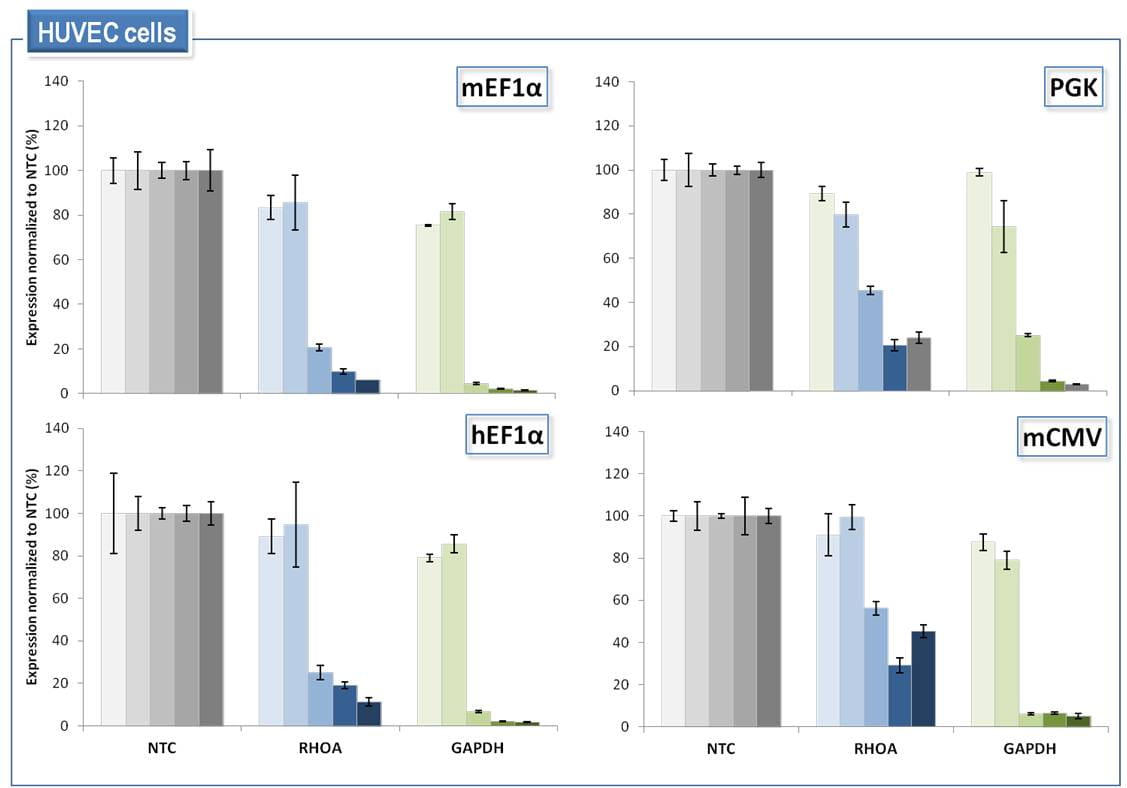
HUVEC(初代ヒト臍帯静脈内皮)細胞に、non-targetingコントロールshRNA(NTC)またはRHOA、GAPDHに対するshRNAを含むSMARTvector Inducible Lentiviral shRNAベクターをMOI = 0.2で形質導入しました。1.5 µg/mLピューロマイシンで72時間細胞を選択しました。次に、shRNAの発現をそれぞれ0、0.01、0.1、1.0、10 µg/mLのドキシサイクリンで誘導し、dox誘導の72時間後にmRNAを回収しました。標的遺伝子ノックダウンは、PPIB参照遺伝子と比較してRT-qPCRによって測定しました。遺伝子ノックダウンは、ベクターおよびドキシサイクリンの投与量が一致したNTC細胞集団と比較して表示しています。
SMARTvector Inducible Lentiviral shRNAは、時間およびドキシサイクリンの用量依存的に必須遺伝子の厳密に制御されたノックダウンを可能にする
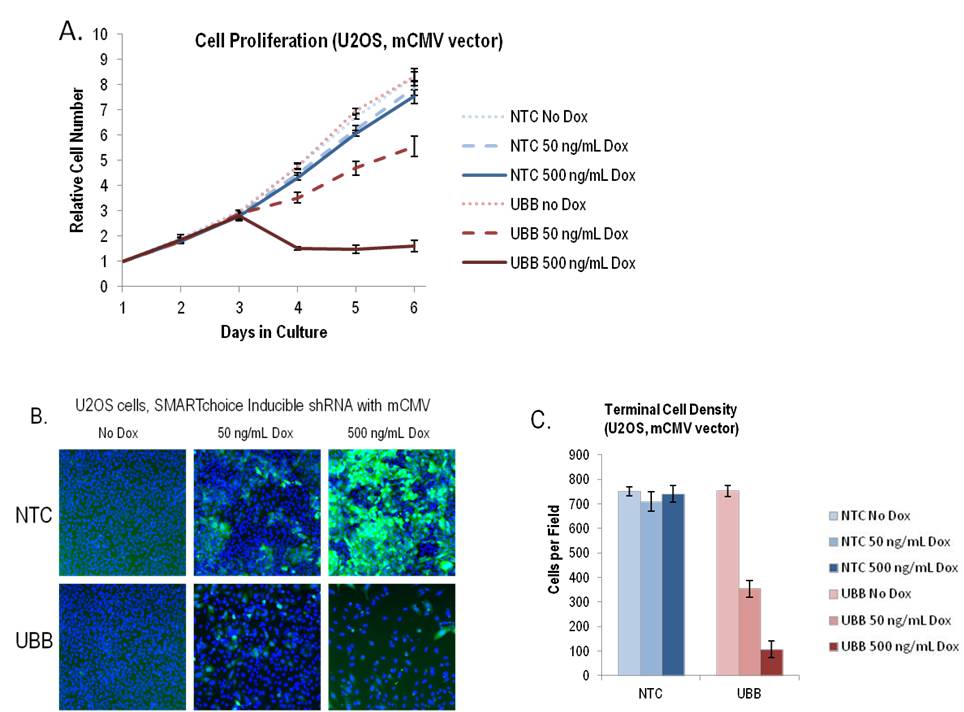
A: U2OS細胞に、non-targetingコントロールshRNA(NTC)またはユビキチンB(UBB)遺伝子に対するshRNAのいずれかを運ぶSMARTvector Inducible Lentiviral shRNAベクター(mCMVプロモーター)をMOI = 0.1で形質導入しました。1.5 µg/mLピューロマイシンで3日間細胞を選択し、選択後、細胞を96ウェルプレートに1ウェルあたり2000細胞で播種しました。24時間後(1日目)、指示された用量のドキシサイクリンでshRNA発現を誘導し、CellTiter-Glo®アッセイ(Promega)で細胞数を毎日測定しました。各データポイントは、6つの独立したウェルの平均と標準偏差を表します。
B: 6日目に、ドキシサイクリンへの5日間の曝露後、細胞をHoescht 33342で染色し、細胞核(青)とTurboGFP(緑)をArrayScan™ VT IHCSリーダーで画像化しました。
C:フィールドあたりの核の数として定義される細胞密度は、条件ごとに18フィールドについて計算しました。
Tet-inducible expression system references:
- Loewr, Heinz N, et al., Improved Tet-responsive promoters with minimized background expression. BMC Biotechnol. 10, 81 (2010).
- Zhou X, Vink M, et al., Optimization of the Tet-On system for regulated gene expression through viral evolution. Gene Ther. 13(19), 1382-1390 (2006).
- Ž. Strezoska, A. Licon, Optimized PCR Conditions and Increased shRNA Fold Representation Improve Reproducibility of Pooled shRNA Screens. PLoS One 7, e42341 (2012).
Product data
Product inserts
Protocols
Safety data sheets
Selection guides
Related Products
Optimized indexed PCR primers for amplification and next-generation sequencing of SMARTvector and shMIMIC pooled libraries
SMARTvector Inducible shRNAを用いた実験の正確なデータ解釈のためのポジティブおよびネガティブコントロール
