- Dharmacon Screening libraries
- Mouse ON-TARGETplus siRNA Library - Druggable Genome
Mouse ON-TARGETplus siRNA Library - Druggable Genome
siRNA designed and modified for greater specificity

The ON-TARGETplus Druggable Genome library contains the genes that are considered potential targets for therapeutics, including kinases, proteases, phosphatases, GPCRs, ion channels, ubiquitin ligases, and genes involved in senescence, autophagy, DNA repair, and characterized nuclear receptors. This extensive library is delivered as nine distinct subset siRNA libraries in the 96-well format (see Supporting Data tab). In the 384-well format, the first eight subsets are plated continuously to reduce partial plates.
The ON-TARGETplus siRNA designs and modifications reduce off-target effects while maintaining high-silencing potency for high-confidence screening results.
Highlights
- Patented dual-strand ON-TARGETplus modification pattern on all siRNAs to reduce off-targets
- Sense strand is modified to prevent interaction with RISC and favor antisense strand uptake
- Antisense strand seed region is modified to destabilize off-target activity and enhance target specificity
- Available as SMARTpool siRNA reagents or a Set of 4 siRNAs in 96 or 384-well plates
- Guaranteed target gene knockdown (see Specifications tab)
Nine subsets make up the mouse druggable genome siRNA library
|
|
Our siRNA knockdown guarantee
siGENOME and ON-TARGETplus siRNA reagents (SMARTpool and three of four individual siRNAs) are guaranteed to silence target gene expression by at least 75% at the mRNA level when demonstrated to have been used under optimal delivery conditions (confirmed using validated positive control and measured at the mRNA level 24 to 48 hours after transfection using 100 nM siRNA).
Note: Most siGENOME and ON-TARGETplus siRNA products are highly functional at 5 to 25 nM working concentration.
The Druggable Genome siRNA library
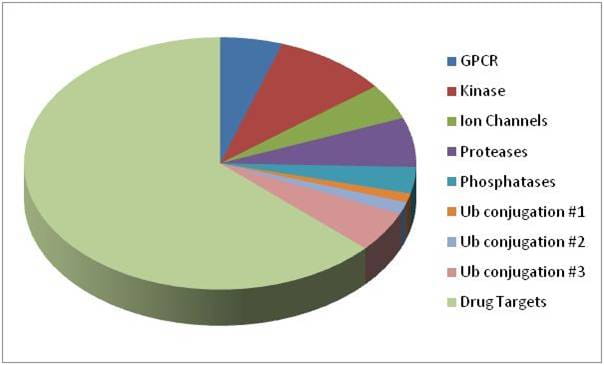
Figure 1. | Pie chart represents the contribution of each gene family collection to the entire Druggable genome. The Drug Targets library (also sold separately) consists of genes involved with apoptosis, senesence, nucleic acid binding, autophagy, DNA repair, and characterized nuclear receptors.
False phenotypes due to off-targets are alleviated by ON-TARGETplus SMARTpool reagents while target gene knockdown is maintained
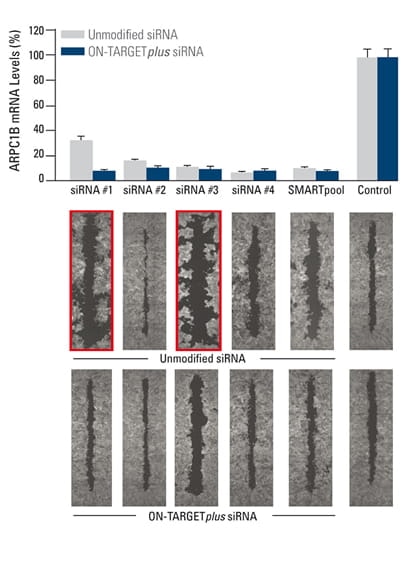
Figure 2. | The effect of silencing ARPC1B on cell migration was studied in a breast cancer cell line. A monolayer of cells was uniformly scraped, and the rate of cell migration to close the scrape (wound healing) was evaluated.
Both unmodified and ON-TARGETplus siRNA reagents induced potent target knockdown. Inconsistent phenotypes due to off-target effects (red outline) were observed for cells transfected with unmodified individual siRNAs. The unmodified SMARTpool improved the false phenotype considerably while the ON-TARGETplus SMARTpool significantly reduced off-target effects to produce a consistent phenotype. In collaboration with Kaylene Simpson, Laura Selfors, and Joan Brugge, Harvard Medical School.
ON-TARGETplus modifications reduce the overall number of off-targets and pooling reduces them even further
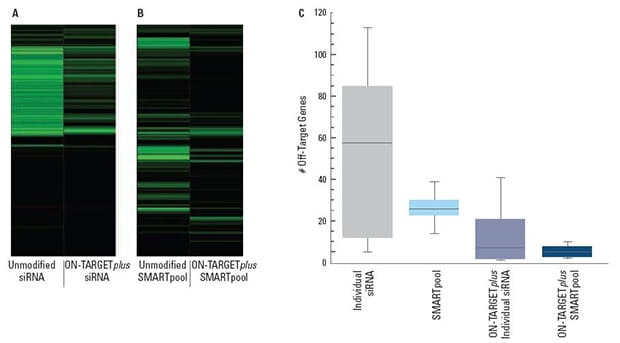
Figure 3. | Panels (A) and (B) are representative examples of off-target signatures with and without application of ON-TARGETplus modifications to (A) a single siRNA and (B) a SMARTpool reagent. Green bars indicate genes with 2-fold or more reduction of expression when treated with the indicated siRNA reagent.
The ON-TARGETplus modifications reduced the off-targets when compared to unmodified siRNA. Pooling of siRNA and the ON-TARGETplus modification pattern independently and in combination, provide significant reduction in off-target gene silencing.
Panel (C) represents quantitation of off-targets (down-regulated by 2-fold or more) induced by the indicated siRNA reagents targeting 10 different genes (4 siRNAs per gene or a single SMARTpool reagent). Off-targets were quantified using microarray analysis (Agilent), then compiled. Each shaded box represents the middle 50% of the data set. Horizontal line in box: Median value of the data set. Vertical bars: minimum and maximum data values.
Product References
- A. L. Hopkins, How many drug targets are there? Nature Reviews Drug Discovery. 5(12), 993-996 (December 2006).
- K. R. Sakharkar, Targetability of human disease genes. Current Drug Discovery Technologies. 4(1), 48-58 (June 2007).
General Screening References
- B. D. Parsons, A. Schindler, D. H. Evans, E. Foley, A direct phenotypic comparison of siRNA pools and multiple individual duplexes in a functional assay. PLoS One. 4(12), e8471 (2009).
- M. Jiang, R. Instrell, B. Saunders, H. Berven, M. Howell, Tales from an academic RNAi screening facility; FAQs. Brief Funct. Genomics. 10(4), 227-237 (2011). [doi: 10.1093/bfgp/elr016]
- J. Borawski, A. Lindeman, F. Buxton, M. Labow, L.A. Gaither, Optimization procedure for small interfering RNA transfection in a 384-well format. J. Biomol. Screen. 12(4), 546-559 (2007).
- T. Ratovitski, E. Chighladze, E. Waldron, R. R. Hirschhorn, C. A. Ross, Cysteine proteases bleomycin hydrolase and cathepsin Z mediate N-terminal proteolysis and toxicity of mutant huntingtin. J. Biol. Chem. 286(14), 12578-12589 (2011). [Human Proteases]
Product inserts
Related Products
Concentrated buffer solution recommended for resuspension and long-term storage of any short, double-strand, or single-strand synthetic RNA molecule. Dilute with RNase-free water prior to use.
Catalog ID:B-002000-UB-100
Unit Size:100 mL
$99.00
Molecular grade water for dilution of 5x siRNA Buffer or resuspension of RNA. RNase-free to prevent degradation of RNA reagents and oligonucleotides.
Catalog ID:B-003000-WB-100
Unit Size:100 mL
$32.00
An aliquot of each of the four DharmaFECT formulations for siRNA/microRNA transfection optimization studies
Catalog ID:T-2005-01
Unit Size:0.2 mL
