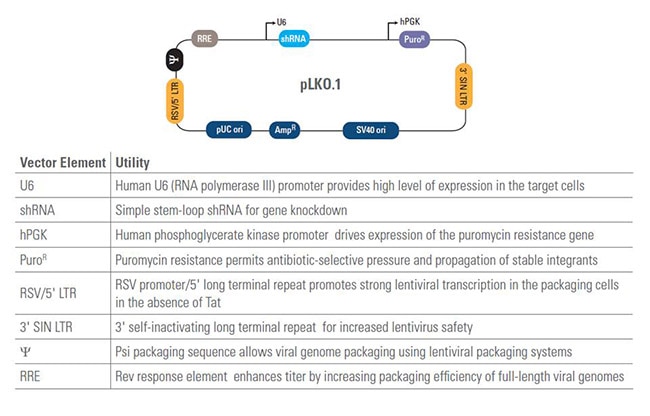
TRC Lentiviral shRNA Controls
TRC positive and negative shRNA controls for a well-designed gene silencing experiment
Positive and negative pLKO.1 shRNA controls are a critical part of a well-designed gene silencing experiment. Available TRC shRNA controls include the pLKO.1 empty vector as a negative control and an eGFP shRNA as a positive control.