- Gene editing
- Pin-point™ Reagents
- Pin-point™ Base Editing mRNA
Pin-point™ Base Editing mRNA
Pin-point base editing mRNAs should be combined with Pin-point validated control sgRNAs, Pin-point Non-targeting control sgRNAs, or Pin-point custom sgRNAs to effectively base edit cells for targeted point mutation or gene knockout without inducing DNA double stranded breaks. Pin-point nCas9 and deaminase mRNAs are available in both Adenine base editor (ABE) and Cytosine base editor (CBE) configurations.

|
Pin-point™ base editing mRNAs harness the DNA targeting ability of a nickase Cas9, with the DNA editing activity of an adenine or cytidine deaminase to introduce targeted point mutations when used in combination with Pin-point synthetic sgRNA. All mRNAs are available with either 5-methoxy-uridine (5moU) base modifications or fully unmodified throughout.
|
Design and order custom CBE or ABE Pin-point sgRNA now
|
Base editing with the Pin-point platform induces specific nucleotide changes without the formation of DNA double-strand breaks or indels.
This system consists of three components:
[1] a nuclease-defective "nickase" nCas9 that only cuts or “nicks” a single strand of DNA. For cytosine base editing (CBE), the nCas9 is fused to a sequence of uracil glycosylase (UGI) inhibitors (Komor, 2016). For adenine base editing (ABE), the nCas9 is not fused to any UGI.
[2] a cytidine deaminase (rAPOBEC) or an adenine deaminase (ABE-flex or ABE-exact) fused to an aptamer binding protein. The rAPOBEC enzyme converts C-G base pairs to T-A base pairs if the base pairs are within the editing window, positions 4 to 8 at the PAM-distal end of the protospacer (Collantes, 2021). ABE-flex converts A-T base pairs to G-C base pairs if the base pairs are within positions 4 to 8 of the base editing window while ABE-exact converts A-T base pairs to G-C base pairs if the base pairs are within positions 5 to 6 of the base editing window.
[3] an aptameric single guide RNA (sgRNA) that recruits the nCas9 and the aptamer-deaminase fusion to a specific DNA target site.
Delivery of all three components into mammalian cells induces the base editing conversion at the targeted site. This system can be used to make gene knockouts through the introduction of premature stop codons (Billon, 2017), the disruption of splice donor and splice acceptor sites (Webber, 2019), or to introduce point mutations. See our Pin-point base editing sgRNA design tool to design your own sgRNAs for gene knockout.
|
|
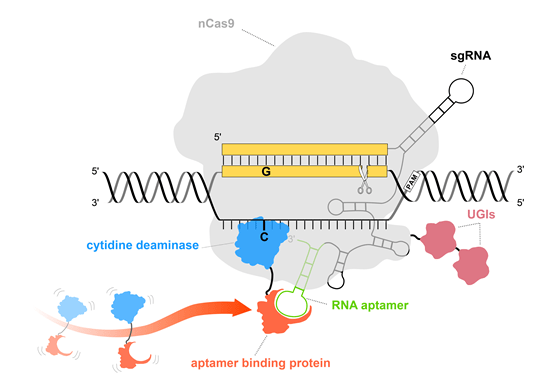
Illustration of Pin-point base editing system.
The utilization of nCas9 (light gray) with or without UGIs (red) ensures that no DNA double-strand breaks occur and DNA damage response pathway is not triggered. Pin-point sgRNA contains an aptamer (green) that is used to recruit deaminase (blue = cytidine deaminase, turquoise = adenine deaminase) via aptamer binding protein (orange) to perform base editing.
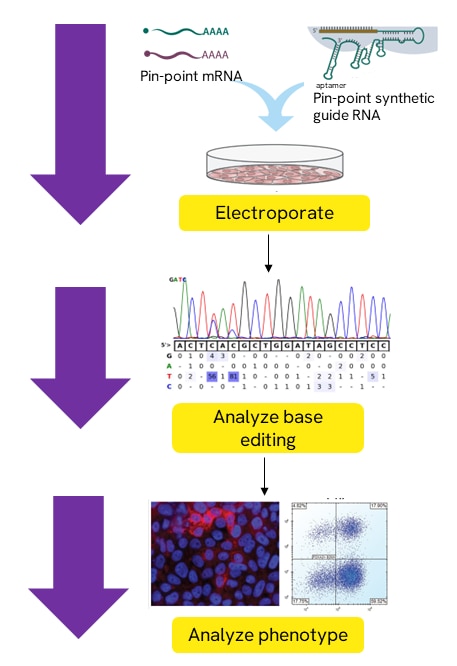
Base editing with Pin-point ABE or CBE nCas9 mRNA, Pin-point ABE or CBE deaminase mRNA, and Pin-point synthetic sgRNA
All three components are electroporated into a specific cells type of interest. Base editing may then be observed using analysis primers to amplify and sequence the base editing region of interest. A phenotype may be analyzed to assess target protein knockout where applicable.
Table 1: Should I select Pin-point unmodified or Pin-point 5moU modified mRNAs?
| Suggested use of Pin-point mRNAs by cell type* | |
| HEK293T cells | unmodified |
| Primary human T cells | unmodified |
| Induced pluripotent stem cells (iPSCs) | unmodified |
| Hematopoietic Stem and Progenitor Cells (HSPCs) | 5moU modified |
Table 2: How much Pin-point ABE mRNA do I need?
| Number of reactions by well/format size* |
|||||||||||
| T cells (250,000 cells per well in a 24 well plate) |
iPSCs (100,000 cells per well in a 12 well plate) |
HSPCs (50,000 cells per well in a 96 well plate) |
|||||||||
| ABE nCas9 mRNA | ABE-exact deaminase mRNA | ABE-flex deaminase mRNA | ABE nCas9 mRNA | ABE-exact deaminase mRNA | ABE-flex deaminase mRNA | ABE nCas9 mRNA | ABE-exact deaminase mRNA | ABE-flex deaminase mRNA |
|||
| 1.5 μg/well | 1 μg/well | 0.1 μg/well | 1 μg/well | 1 μg/well | 0.1 μg/well | 1.5 μg/well |
1 μg/well | 0.4 μg/well | |||
| 10 μg (2 μg/μL) | - | 10 | 100 | - | 10 | 100 | - | 10 | 25 | ||
| 20 μg (2 μg/μL) | 13 | 20 | 200 | 20 | 20 | 200 | 13 | 20 | 50 | ||
| 100 μg (2 μg/μL) | 67 | 100 | 1000 | 100 | 100 | 1000 | 67 | 100 | 250 |
||
| 500 μg (2 μg/μL) | 333 | - | 500 | - | - | 333 | - | - |
|||
Table 3: How much Pin-point CBE mRNA do I need?
| Number of reactions by well/format size* | ||||||||
| T cells (250,000 cells per well in a 24 well plate) |
iPSCs (100,000 cells per well in a 12 well plate) |
HSPCs (50,000 cells per well in a 96 well plate) |
HEK 293T cells (50,000 cells per well in a 96 well plate) |
|||||
| CBE nCas9 mRNA |
CBE rAPOBEC deaminase mRNA | CBE nCas9 mRNA | CBE rAPOBEC deaminase mRNA | CBE nCas9 mRNA | CBE rAPOBEC deaminase mRNA | CBE nCas9 mRNA | CBE rAPOBEC deaminase mRNA | |
| 1.56 μg/well |
0.22 μg/well
|
2.56 μg/well | 0.4 μg/well | 2.8 μg/well | 2 μg/well | 1 μg/well | 0.1 μg/well | |
| 20 μg (2 μg/μL) |
12 | 90 | 8 | 50 | 7 | 10 | 20 | 200 |
| 100 μg (2 μg/μL) | 64 | 450 | 39 | 250 | 36 | 50 | 100 | 1000 |
| 500 μg (2 μg/μL) |
320 | 2252 | 195 | 1250 | 179 | 250 | 500 | 5000 |
Table 4: How much Pin-point sgRNA do I need?
| Number of reactions by well/format size* | ||||||||
| T cells (250,000 cells per well in a 24 well plate) |
iPSCs (100,000 cells per well in a 12 well plate) |
HSPCs (50,000 cells per well in a 96 well plate) |
HEK 293T cells (50,000 cells per well in a 96 well plate) |
|||||
| CBE |
ABE | CBE | ABE | CBE | ABE | CBE | ||
| 20 pmol/well |
20 pmol/well
|
40 pmol/well | 40 pmol/well | 62.5 pmol/well | 62.5 pmol/well | 60 pmol/well | ||
| 2 nmol | 100 | 100 | 50 | 50 | 32 | 32 | 33 | |
| 5 nmol | 250 | 250 | 125 | 125 | 80 | 80 | 83 | |
| 10 nmol | 500 | 500 | 250 | 250 | 160 | 160 | 167 | |
For additional Supporting Data please see our Pin-point Synthetic sgRNA Validated Controls, Pin-point Synthetic sgRNA Non-targeting Controls, or Custom Pin-point Synthetic sgRNA pages

Figure 1: Efficient base editing in activated human T cells with the Pin-point ABE platform at CD52 locus.
(A) CD52 sgRNA spacer sequence. Numbers indicate positions within spacer. A6 is on-target for generating splice donor mutation and protein knockout; other A positions are bystanders in this example. (B-C) Base editing levels following mRNA titrations of Pin-point ABE-exact (B) and ABE-flex (C) at on-target A (6) and the bystander A (12). Amounts of Pin-point ABE nCas9 mRNA and synthetic sgRNA were kept constant. Percent A to G conversion was calculated from Sanger sequencing of the PCR products using our Pin-point analysis primers. N=2 technical replicates. Points – average ± SD.

Figure 2: Efficient base editing in hematopoietic stem and progenitor cells (HSPCs) with the Pin-point ABE platform at HBG1/2 promoter regions.
(A) HBG1/2 sgRNA spacer sequence. A5 and A8 fall within BCL11A binding site. Other A positions are bystander. (B) Base editing levels following editing with nCas9 ABE mRNA, ABE-exact or ABE-flex deaminase mRNAs, and HBG1/2 synthetic sgRNA control. Bars – average ± SD. N=2 technical replicates. (C) Base editing levels following titration of Pin-point ABE-flex mRNA in HSPCs. Amounts of Pin-point ABE nCas9 mRNA and synthetic sgRNA were kept constant. Percent A to G conversion was calculated from Sanger sequencing of the PCR products using our Pin-point analysis primers. N=2 technical replicates. Bars and points – average ± SD.

Figure 3: Evaluating the effects of Pin-point ABE mRNA modification on base editing levels.
(A) Unmodified or 5moU-modified ABE nCas9 mRNA and ABE-exact deaminase mRNA were delivered to activated human T cells via electroporation, together with Pin-point CD52 synthetic sgRNA ABE validated control. (B) Unmodified or 5moU-modified ABE nCas9 mRNA and ABE-flex deaminase mRNA were co-delivered with Pin-point CD52 synthetic sgRNA ABE validated control to activated human T cells via electroporation. (C) Unmodified or 5moU-modified ABE nCas9 mRNA and ABE-flex deaminase mRNA were co-delivered with Pin-point HBG1/2 synthetic sgRNA ABE validated control to HSPCs via electroporation. Percent A to G conversion was calculated from Sanger sequencing of the PCR products using our Pin-point analysis primers. N=2 technical replicates. Bars – average ± SD.

Figure 4: Pin-point CBE base editing using unmodified or 5moU modified mRNAs in T cells, HSPCs, and induced pluripotent stem cells (iPSCs).
Representative editing(A) and total cell count at 3 days post electroporation (B) when delivering a custom Pin-point synthetic sgRNA targeting a chain of the major histocompatibility complex class 1 and either Pin-point unmodified or Pin-point 5moU modified CBE mRNAs in activated human T cells, iPSCs, and HSPCs.

Figure 5: Efficient multiplex base editing with the Pin-point CBE platform in human T cells.
Activated human T cells were electroporated with Pin-point CBE mRNAs and Pin-point synthetic sgRNA CBE validated controls for the 3 targets (CD52, PDCD1 and TRAC), or non-targeting control (NTC #1) sgRNA. Mock-electroporated (EP) cells were used as negative controls. (A) Cells were harvested 6-7 days post electroporation, and base editing levels were calculated from Sanger sequencing data of PCR products using the Pin-point analysis primers. N = 8 T cell donors, 5 independent experiments with duplicate or triplicate technical replicates. Bars are averages ± SD. (B) Cells were analyzed by flow cytometry 7 days post electroporation. To induce the expression of PD1, T cells were cultured in the presence of phorbol 12- myristate 13-acetate and ionomycin for 48 h prior. Expression of CD52 and TCRa/b were analyzed on non-stimulated cells. Percentage of negative cells for each marker in the edited and mock-electroporated (EP) reported as percentage of live cells. N = 2 T cell donors with duplicate or triplicate technical replicates. Bars are averages ± SD.
Application notes
Posters
-
Performance and modularity of Revvity's Pin-point base editing system characterized by arrayed and pooled screening platforms - poster
-
Pin-point™ base editing system: a versatile editing platform driving cell therapies - poster
-
The Pin-point™ base editing platform streamlines the generation of hypoimmunogenic iPSCs for allogeneic cell therapy - poster
Safety data sheets
Selection guides
Short protocols
-
Pin-point™ CBE base editing in HEK293T and T cells, Neon Electroporation – Short Protocol
-
Pin-point™ ABE base editing in HPSCs – Short Protocol
-
Pin-point™ ABE base editing in iPSCs – Short Protocol
-
Pin-point™ ABE base editing in T cells – Short Protocol
-
Pin-point™ CBE base editing in HEK293T and T cells, Lonza 4D-Nucleofection – Short Protocol
-
Pin-point™ CBE base editing in HPSCs – Short Protocol
-
Pin-point™ CBE base editing in iPSCs – Short Protocol
Related Products
Revvity’s Pin-point™ base editing design tool makes it simple to create base editing sgRNAs that deliver reliable, high-efficiency gene knockouts.
Non-targeting synthetic controls for evaluation of Pin-Point base editing. Bioinformatically designed to not target any gene in the human genome
Synthetic sgRNA controls to verify optimal parameters for precise gene editing without inducing DNA double strand breaks

