- Gene editing
- Gene editing reagents
- Edit-R All-in-one lentiviral sgRNA negative controls
Edit-R All-in-one lentiviral sgRNA negative controls
All-in-one non-targeting constructs are bioinformatically designed and validated to not target any gene in human or mouse genomes

AAVS1/Rosa26 cutting controls are designed to target regions of the genome that do not result in functional knockout of a protein or known phenotypic changes. These regions are sometimes known as safe harbor regions. These guides are validated for mismatch detection assays and are recommended for screening applications as an additional control that results in Cas9 cutting without a phenotypic readout.
Non-targeting controls are recommended as negative controls for experiments using lentiviral sgRNA. All Edit-R non-targeting controls are designed to have a minimum of three mismatches or gaps to all potential PAM-adjacent targets in the human or mouse genomes. Changes in viability or gene expression levels in cells treated with these controls likely reflect a baseline cellular response that can be compared to the levels in cells treated with target-specific sgRNAs.
Edit-R CRISPR-Cas9 Gene Engineering Platform
The Dharmacon Edit-R Gene Engineering platform is powered by a proprietary algorithm and based on the Type II CRISPR-Cas9 system from the bacteria Streptococcus pyogenes.
- Ready-to-Use Components: The system eliminates the need for cloning, allowing researchers to quickly initiate experiments with predesigned guide RNAs and other essential components, significantly reducing the time to results.
- High Specificity and Efficiency: Edit-R utilizes a proprietary algorithm to design guide RNAs that improve the likelihood of achieving functional knockouts while minimizing off-target effects, ensuring high confidence in experimental outcomes.
- Guaranteed Performance: Every predesigned guide RNA is backed by a guarantee of successful editing at the target site, providing researchers with additional assurance in their gene editing endeavors.
The Edit-R predesigned guide RNA guarantee
We guarantee that EVERY predesigned guide RNA will provide successful editing at the target site when delivered as described in the Edit-R Technical Manuals.
The Edit-R guide RNA guarantee is valid when used with any wild type S. pyogenes Cas9 nuclease, including mRNA, expression plasmid, protein, or stable Cas9 expression, and Edit-R crRNAs must be used with Edit-R tracrRNA for the guarantee to apply.
Analysis of editing of the treated cell population must be shown using a T7EI or Surveyor mismatch detection assay. If successful editing is not observed for a predesigned Edit-R guide RNA while an appropriate side-by-side Edit-R positive control is successful, a one-time replacement of a different predesigned Edit-R guide RNA of the same format and quantity will be provided at no cost.
A replacement will only be approved upon discussion with our Scientific Support team.
Successful editing at the DNA level does not always lead to functional gene knockout; it is recommended to test multiple guide RNAs to determine the most effective guide RNA for knockout of your target gene.
This guarantee does not extend to any accompanying experimental costs, does not apply to guide RNAs ordered via the CRISPR Design Tool, and will not be extended to the replacement guide RNA.
Gene knockout workflow using the Edit-R All-in-one lentiviral sgRNA system
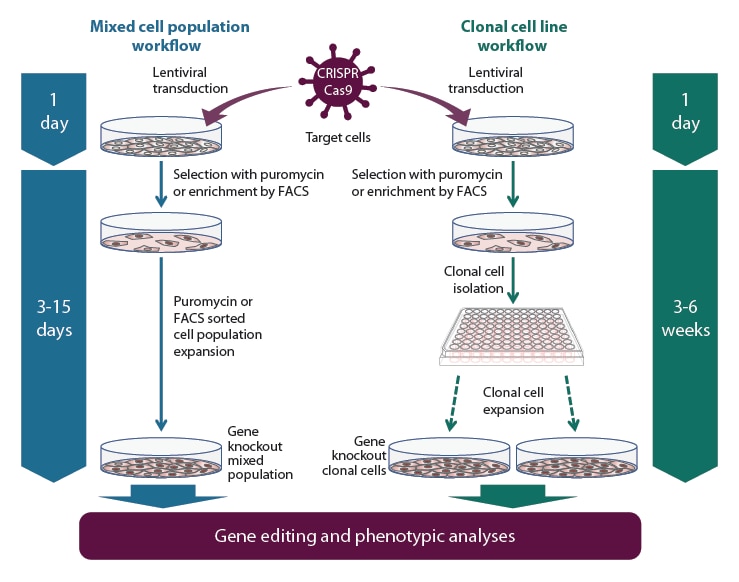
Gene knockout workflow with the All-in-one lentiviral sgRNA using a mixed popultion (left side) or clonal cell line (right side) experimental approach.
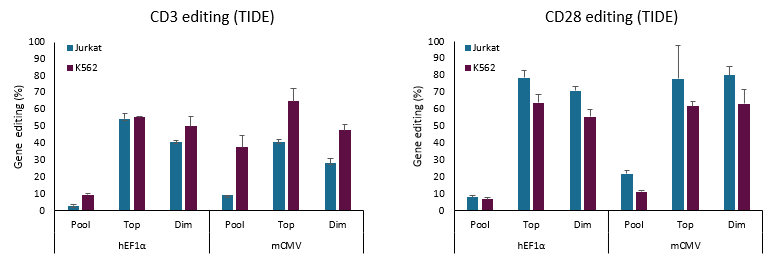
Edit-R All-in-one lentiviral system percent editing (TIDE)
Jurkat or K562 cells were transduced at a MOI of 0.3 using Edit-R All-in-one EGFP Lentiviral sgRNA vectors targeting either the CD3 or CD28 genes. All-in-one lentiviral particles express Cas9 nuclease constitutively under the hEF1α or mCMV promoters. Lentiviral particles expressing Non-targeting control (NTC) guides were used as negative controls. After lentiviral transduction and expansion, cells were analyzed as unsorted population (pool) or were subjected to cell sorting to enrich for the brightest (top) or dimmest (dim) EGFP expressing cell populations. The pool, top, and dim populations of cells were then subjected to TIDE analysis to assess gene editing frequency.
Comparison of Edit-R two-vector and All-in-one systems
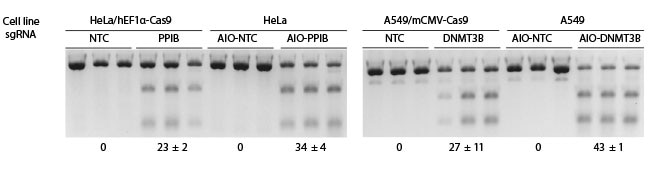
For the two-vector system, cells were previously transduced with a constitutive hEF1α- or mCMV-Cas9 expression lentiviral particles at an MOI of 0.3, and selected with 10 µg/mL blasticidin for 10 days, followed by transduction with non-targeting control (NTC), DNMT3B- or PPIB-sgRNA lentiviral particles at an MOI of 0.3. For the All-in-one system, wild-type cells were transduced with the All-in-one were selected with 2 µg/mL puromycin for 3 days. For the All-in-one system, wild-type cells were transduced with the NTC, DNMT3B or PPIB All-in-one lentiviral particles that also express a constitutive hEF1α- or mCMV-Cas9 nuclease, and the PuroR gene at an MOI of 0.3. All transduced cells were selected with 2 µg/mL puromycin for 3 days. The cells were then lysed and analyzed for indels using a DNA mismatch detection assay with T7EI.
Schematic diagram of the Edit-R All-in-one lentiviral sgRNA vector
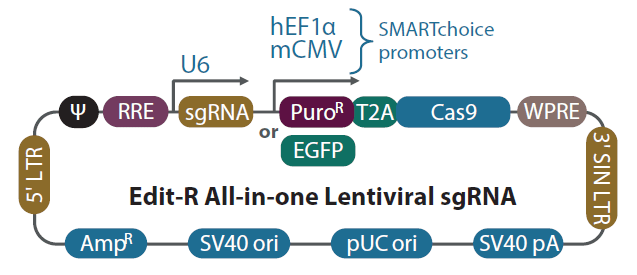
In the Edit-R All-in-one lentiviral sgRNA vector backbone, the gene-specific guide RNA is expressed under the control of a human U6 promoter, while expression of the Cas9 and puromycin resistance marker (PuroR) or EGFP marker is driven from either the human EF1α or the mouse CMV promoter. The plasmid contains the AmpR resistance marker for growth and selection in E. coli.
| Vector Element | Utility |
|---|---|
| Cas9 | Human codon-optimized S. pyogenes Cas9 nuclease for cleavage of targeted DNA when programmed with a sgRNA |
| T2A | Self-cleaving peptide allows for simultaneous expression of puromycin resistance and Cas9 protein from a single transcript |
| PuroR | Puromycin resistance marker permits antibiotic selection of transduced mammalian cells |
| EGFP | Fluorescent marker permits FACS selection of transduced mammalian cells |
| mCMV | Mouse cytomegalovirus immediate early promoter |
| hEF1α | Human elongation factor 1 alpha short promoter |
| U6 | Human RNA polymerase III promoter U6 |
| sgRNA | Optimized single guide RNA, a fusion of gene-specific crRNA with the tracrRNA scaffold |
| 5' LTR | 5' Long Terminal Repeat necessary for lentiviral particle production and integration of the construct into the host cell genome |
| Ψ | Psi packaging sequence allows lentiviral genome packaging using lentiviral packaging systems |
| RRE | Rev Response Element enhances titer by increasing packaging efficiency of full-length lentiviral genomes |
| WPRE | Woodchuck Hepatitis Post-transcriptional Regulatory Element enhances transgene expression in target cells |
| 3' SIN LTR | 3' Self-inactivating Long Terminal Repeat for generation of replication-incompetent lentiviral particles |
| SV40 pA | Simian virus 40 polyadenylation signal |
| pUC ori | pUC origin of replication |
| SV40 ori | Simian virus 40 origin of replication |
| AmpR | Ampicillin resistance gene for vector propagation in E. coli cultures |
Product inserts
Protocols
Safety data sheets
Related Products
Combine sgRNA and Cas9 nuclease expression into a single vector for simplified delivery
Controls to verify DNA double-strand breaks and gene editing efficiencies