
遺伝子改変細胞株作製プロジェクトの規模や複雑さに関わらず、当社はお客様の創薬研究をスピーディーに進めるよう支援いたします。遺伝子改変細胞株の作製は、新しい可能性の世界を切り開きました。オートメーションによる規模拡大、および習熟したサイエンティストによる複雑な改変をもとに、当社は様々な遺伝子改変細胞株を提供しています。遺伝子改変細胞株作製の当社への委託で、お客様は細胞株作製後のご研究にご注力いただけます。
あらゆる細胞株のあらゆる遺伝子改変において当社をぜひご活用下さい。
When it’s time to take your research to the next level, Horizon Discovery is there for you.
複雑な遺伝子改変を取り組む専門性
- 10年以上の遺伝子改変細胞株作製の経験
- 数百の細胞株にわたる数千の改変の実績
- 課題が顕在化する以前での解決に向けた科学的コンサルテーション
ご要望に柔軟でタイムリーに対応します。
- シンプルな改変から複雑な改変まで
- オートメーションによる大型プロジェクトの発注可
- ライセンス確保済の数百の細胞株から選択可(Licensingタブ参照)。お客様保有の細胞株の編集も可。
- プールまたはクローンをご選択可(プロジェクトに依存)。
バイオロジー研究に集中できる安心をご提供します。
- 包括的なデータパッケージ
- シーケンスバリデーション
- マイコプラズマ試験/無菌試験
低リスク
- 遺伝子改変細胞が取得できない場合ご請求いたしません(成功報酬型。ただしプロジェクトに依存)。

Project options
- シングルノックアウト
- ダブルノックアウト
- 小規模欠失
- 大規模ゲノム欠失
- 点変異(SNP)ノックイン
- 接合体の指定が可能(要追加費用)
- タグ/レポーターノックイン
- Turbo GFP(Evrogen社)、Turbo RFP(Evrogen社)
- His、Myc、またはFLAG
- HiBiT(プロメガ社)、LgBiT(プロメガ社)
- NanoLUC®:Protein Reporter(プロメガ社)、またはNanoLUC®:Promoter Reporter(プロメガ社)
- N末端またはC末端の挿入位置へのノックイン
- 大規模ノックイン
- iPS細胞編集
- シングルノックアウト
- 点変異(SNP)ノックイン
- タグ/レポーターノックイン
- 大規模ノックイン
- ゲノム編集の困難な細胞株
- ランダムインテグレーション– Cas9、過剰発現など
- CRISPRを介したゲノム転座
- より複雑な改変:大規模ノックイン、大規模欠失など
- ご希望のオプションが見つからない場合は、お問い合わせください。Horizonは、様々な細胞株でほぼすべての遺伝子改変を実施するために必要な蓄積された知識と専門性を保有しています。
作製ワークフロー
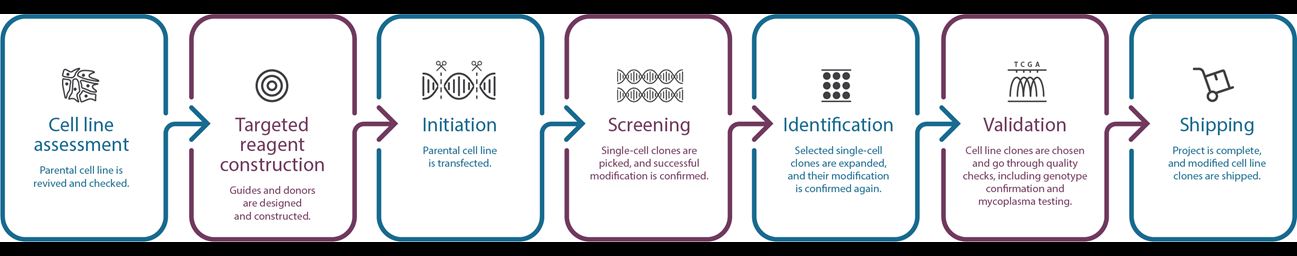
-
追加料金不要で、お客様保有の細胞株の遺伝子改変細胞株の作製が可能です。オンボーディングプロセス(お客様保有の細胞株の当社評価)では、マイコプラズマ試験および無菌試験の実施、およびトランスフェクション条件検討やシングル細胞限界希釈での細胞増殖の評価を行います。
-
細胞株の評価フェーズと並行して、作製工程で使用する試薬の設計・評価を行います。
-
遺伝子改変細胞株作製のフェーズでは、作業明細書(Statement of Work: SoW)で定義されたお客様ご所望の遺伝子改変を実施します。プロセス全体を通じて、プロジェクトマネージメントチームが定期的に進捗状況をお知らせし、作製プロセスで課題が発生した場合等はすみやかにご連絡致します。
-
作製作業完了後、目的の遺伝子改変についてバリデーションを実施します。通常は、サンガーシーケンシングを活用しゲノムレベルのバリデーションを行い、レポーター細胞株についてはレポーター発現データを提供します。
Licensing and cell lines
Horizonは、広範な親株細胞株での改変を実施するために、複数のライセンサーと契約を結んでいます。さらに、クライアントの皆様の他のプロバイダーからの細胞株の入手をサポートできる場合があります。
ノックイン
* CRISPR Cas9システムと方法論を利用して遺伝子をノックインした製品、およびそれらの使用は、Sigma-Aldrich Co. LLC(SIGMA)によって管理されている次の特許およびアプリケーションの1つ以上の対象となる場合があります。Patent applications and issued patents that entered their respective National Stage from PCT International Pub. No. WO 2014/089290, including, but not limited to, the following, and substitutions, divisions, continuations, continuations-in-part, reissues, re-examinations, and extensions thereof: Australia Patent Nos. 2013355214; 2017204031; and 2018229489; Canada Patent Nos. 2,891,347 and 2,977,152; China Patent No. CN105142669; European Patent Nos. EP 2 928 496 B1; EP 3 138 910 B1, 3 138 911 B1, EP 3 138 912 B1, EP 3 360 964 B1, EP 3 363 902 B1; Israel Patent No. IL238856; Singapore Patent No. 11201503824S; South Korea Patent Nos. 10-1844123 and 10-2006880; and U.S. Patent Nos. 10,731,181 and 10,745,716; and U.S. Application Serial Nos. 15/188,927; 15/188,931; and 15/456,204 (the “Patent Rights”).
iPSC編集
in vitroで分化した疾患モデルを利用する特定の薬物スクリーニング、標的検証、および毒性試験方法は、Evotec International GmbHが管理する1つ以上の特許の対象となる場合があります。これらの特許には、EP2270196B1、EP1745144B1、EP3070174B1、および対応する外国特許と係属中の出願が含まれます。この製品の購入または譲渡は、明示的または黙示的を問わず、前述の特許に基づいて実施する権利またはライセンスを付与することを意図したものではありません。そのような特許に基づいて実施するライセンスに関する情報については、Evotec International GmbH(info@evotec.com)に連絡し、入手することをお勧めします。
*誘導性細胞株プロジェクトに関するご留意 Horizonは、誘導性のカスタム遺伝子改変細胞株作製にTet-On 3G誘導性発現システムを使用しています。このプラットフォームは、Tet-On®3G二分割誘導システムに基づいています。これは、最適化された誘導性RNAポリメラーゼIIプロモーター(TRE3G)で構成される厳密に制御されたシステムであり、ドキシサイクリンによる誘導時に最小限の基礎発現と強力な活性化の両方を提供します。誘導性遺伝子改変カスタム細胞株作製プロジェクトを開始する前に、このシステムを自由に使用するため、お客様の責任でClontech Laboratoriesからライセンスを入手する必要があります。
販売条件
すべての遺伝子改変細胞株作製サービスは、当社の販売条件および限定使用ラベルライセンス(LULL: LIMITED USE LABEL LICENSE AGREEMENT)の対象となります。


